Adverse Events Following Immunization
An adverse event following immunization (AEFI) is an unwanted or unexpected health outcome that happens after someone receives a vaccine, which may or may not be caused by the vaccine. Although rare, AEFI’s can occur. Potential AEFI side effects and management should be discussed with the Health Care Provider immunizing you prior to immunization.
AEFI Management
If you believe you or your child is experiencing an adverse event following immunization, it is important to seek a medical evaluation from a health care professional to allow for appropriate treatment and reporting. Depending on severity, this can be done through your family doctor or nurse practitioner, a walk-in clinic or the local emergency department.
AEFI Reporting
Health care providers (i.e., physicians, nurses and pharmacists) are required under the Health Protection and Promotion Act (HPPA) Section 38 and Reg. 569 to report AEFI’s to their local health unit in order to:
- Identify and investigate serious or unexpected occurrences of AEFIs, particularly for new vaccines.
- Detect and investigate safety signals (e.g., lot specific problems)
- Estimate provincial rates of reported AEFI by vaccine
- Report to stakeholders on the safety of publicly funded vaccines in Ontario
- Maintain public confidence in vaccine programs
TBDHU’s Role in AEFI Reporting
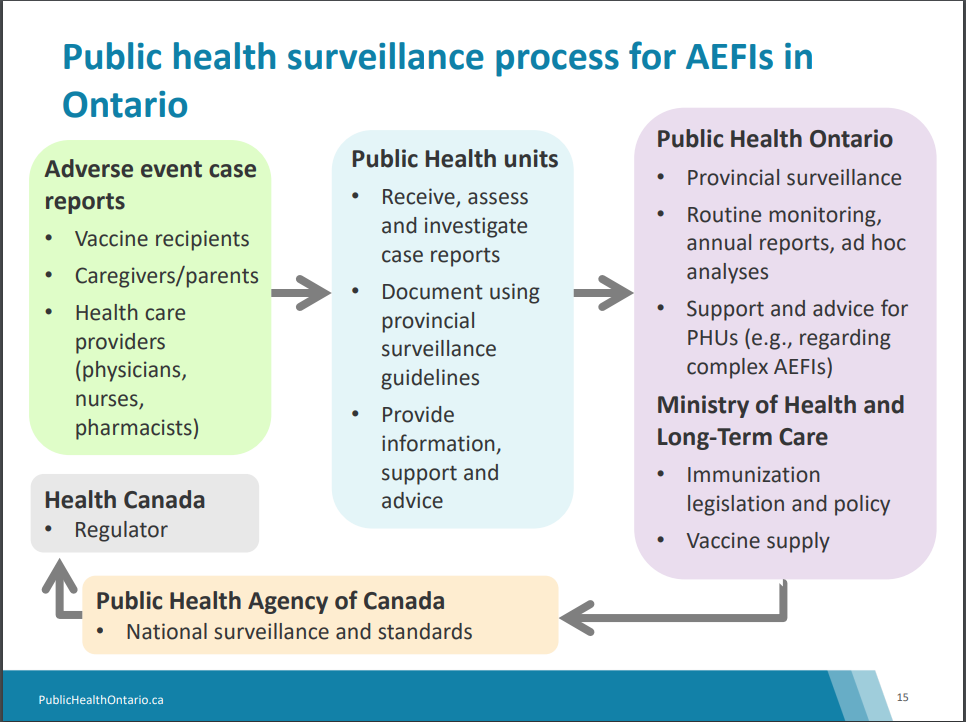
Upon receiving an AEFI report from a Health Care professional, a nursing member of the vaccine preventable disease team will contact the individual or parent/guardian to discuss the AEFI further. Nursing staff can also report an AEFI following a conversation with a vaccine recipient or parent/guardian. At this time, further details and information is collected for assessment and investigation. Once all required information is collected, AEFIs that meet the confirmed case definition are reported to Public Health Ontario then onto the Canadian Adverse Events Following Immunization Surveillance System (CAEFISS) at the Public Health Agency of Canada. Refer to the image below for a complete overview of the process.
For Further Information
Contact the Vaccine Preventable Diseases Program at (807) 625-5900.